







The implementation of an ion exchange resin column represents a cornerstone of modern chemical engineering, providing a highly efficient method for the selective removal of ions from aqueous solutions. From purifying ultra-pure water for semiconductor fabrication to recovering precious metals in hydrometallurgy, these columns serve as the physical vessel where complex chemical swaps occur with surgical precision. Understanding the synergy between the column design and the specific resin chemistry is essential for optimizing throughput and ensuring the longevity of the system.
Globally, the demand for high-performance water treatment and specialty chemical purification has surged, making the ion exchange resin column more relevant than ever. As industrial regulations regarding wastewater discharge become more stringent, industries are shifting toward closed-loop systems where resins are not just filters, but strategic tools for resource recovery. This evolution transforms the column from a simple utility into a value-generating asset within the production line.
For operators and engineers, the key to success lies in mastering the balance of flow rates, bed depth, and regeneration cycles. A poorly optimized ion exchange resin column can lead to premature breakthrough, increased chemical waste, and inconsistent product quality. By leveraging advanced resin types—such as chelating or mixed bed resins—companies can achieve purity levels that were previously thought impossible, driving innovation across the pharmaceutical and energy sectors.

The global market for water purification and ion exchange technologies has seen steady growth, driven by ISO standards and the World Bank's focus on clean water access. The ion exchange resin column is at the heart of this growth, serving as the primary technology for removing dissolved salts and contaminants from industrial streams. As emerging economies scale up their chemical manufacturing, the need for precise ion control has shifted from a luxury to a regulatory requirement.
However, many industries face the challenge of "resin fouling" and high regeneration costs. Traditional columns often suffer from uneven flow distribution, leading to "channeling" where the liquid bypasses the resin beads, significantly reducing the efficiency of the process. This industrial bottleneck has pushed the development of smarter column geometries and high-capacity resins to maintain consistent purity levels in high-volume environments.
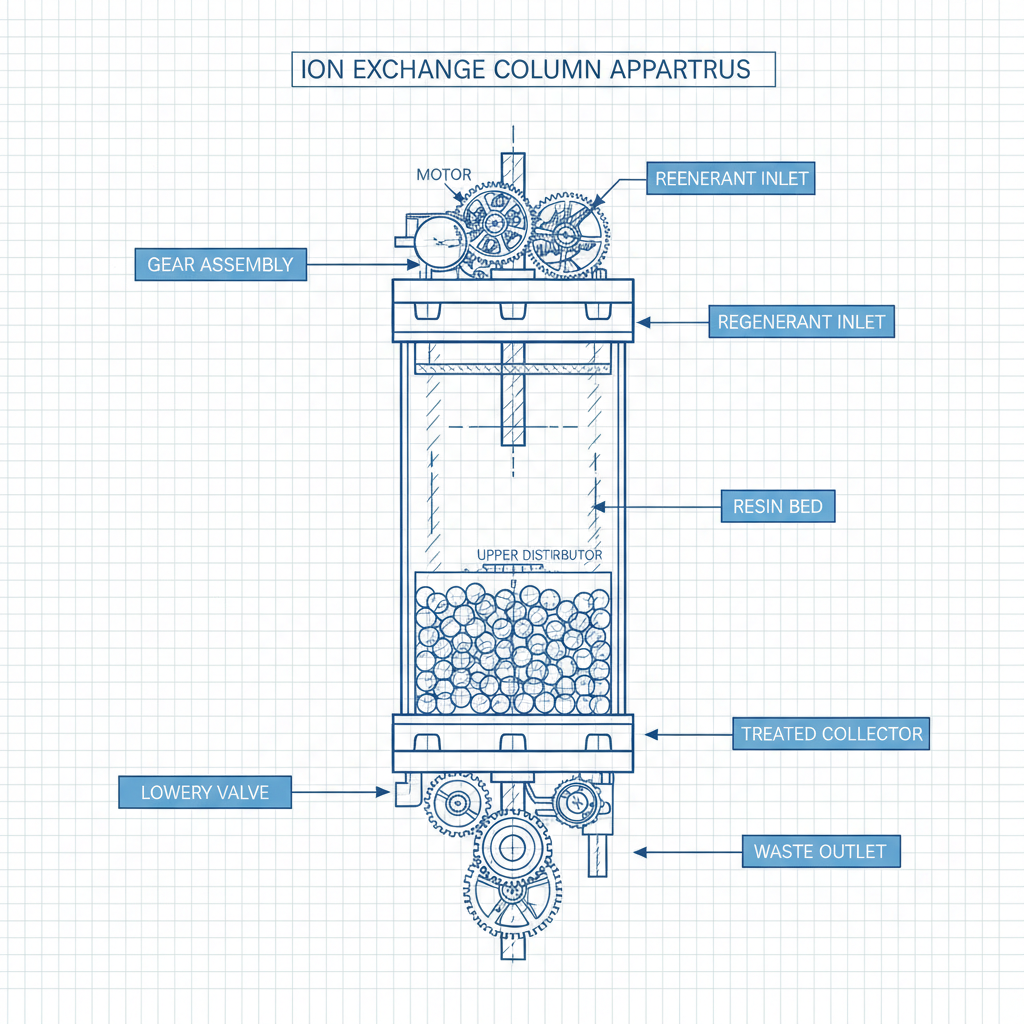
At its simplest, an ion exchange resin column is a pressurized vessel containing a bed of spherical polymer beads that possess functional groups capable of swapping ions with a passing liquid. When a contaminated solution flows through the column, target ions (such as calcium, magnesium, or heavy metals) adhere to the resin, while harmless ions (like sodium or hydrogen) are released back into the stream.
This process is not merely a filtration technique but a sophisticated chemical equilibrium. The effectiveness of the column depends on the affinity of the resin for specific ions. For example, a strong acid cation exchange resin in a column will aggressively target polyvalent metal ions, making it indispensable for water softening and the initial stages of metal recovery.
In modern industry, these columns are integrated into complex trains of treatment. Whether used in a "lead-lag" configuration to prevent breakthrough or as a single polishing unit, the ion exchange resin column ensures that the final effluent meets the precise chemical specifications required for high-tech manufacturing or humanitarian water safety.
The physical integrity of the ion exchange resin column starts with the vessel material. Depending on the chemicals used for regeneration—such as strong hydrochloric acid or sodium hydroxide—columns are typically constructed from FRP (Fiber Reinforced Plastic), stainless steel, or rubber-lined carbon steel to prevent corrosion and leakage.
Internal distribution systems are perhaps the most critical yet overlooked part of an ion exchange resin column. Properly designed headers and laterals ensure that the fluid enters the resin bed uniformly, avoiding dead zones and ensuring that every single resin bead is utilized to its maximum capacity before the column reaches saturation.
Finally, the choice of the resin itself—whether it be a Chelating Resin for heavy metals or a Mixed Bed Resin for ultra-pure water—defines the column's purpose. The interaction between the resin's particle size and the column's pressure drop determines the energy efficiency of the pumping system and the overall operational cost of the installation.
When scaling an ion exchange resin column from a laboratory pilot to an industrial plant, the Aspect Ratio (height-to-diameter) becomes the primary design variable. A tall, narrow column provides a longer contact time (Empty Bed Contact Time - EBCT), which is essential for slow-reacting ions, whereas a wider column allows for higher flow rates with lower pressure drops.
Efficiency is also measured by the "regeneration efficiency," which is the amount of chemical required to restore the resin's capacity. Modern ion exchange resin column systems now employ counter-current regeneration, where the regenerant flows in the opposite direction of the service flow, resulting in significantly lower chemical consumption and higher treated water quality.
The versatility of the ion exchange resin column allows it to operate in vastly different environments. In the pharmaceutical industry, these columns are used for the purification of proteins and the removal of endotoxins, where high-grade Special Resin is required to ensure patient safety and drug efficacy.
In remote industrial zones, such as mining sites in South America or Africa, hydrometallurgy resin columns are used to extract gold, copper, and uranium from lean ores. These installations often operate in harsh conditions, requiring robust column designs that can withstand extreme temperature fluctuations and corrosive leach liquors while maintaining high selectivity for the target metal.
Investing in a high-quality ion exchange resin column provides tangible long-term value through the reduction of operational expenses. By optimizing the resin bed and using high-capacity Weak Base Anion Exchange Resins, plants can extend the time between regeneration cycles, reducing the volume of chemical waste generated and lowering the overall carbon footprint of the facility.
Beyond the financial metrics, there is a significant social impact. The use of these columns in municipal water treatment ensures that communities have access to water free from harmful nitrates and heavy metals. This promotes public health and dignity, providing a reliable foundation for urban development in water-stressed regions.
Moreover, the move toward "circular chemistry" means that an ion exchange resin column is no longer seen as a waste-generator but as a recovery tool. The ability to capture and reuse rare earth elements from industrial waste streams transforms a potential environmental hazard into a sustainable source of raw materials.
The next generation of the ion exchange resin column is being shaped by the "Industry 4.0" revolution. Digital transformation is introducing real-time sensors that monitor conductivity and ion concentration at multiple points within the resin bed. This allows for "predictive regeneration," where the column is regenerated only when absolutely necessary, eliminating the waste associated with fixed-timer schedules.
Material science is also evolving, with the development of nano-composite resins that offer higher exchange capacities and faster kinetics. These new materials allow for the design of smaller, more compact columns that can achieve the same results as traditional large-scale vessels, reducing the physical footprint of industrial plants and lowering construction costs.
Finally, the integration of green energy is seeing the rise of solar-powered regeneration systems in remote areas. By combining automated ion exchange resin column technology with renewable energy, we are moving toward a future where high-purity water and mineral recovery are decoupled from fossil fuel dependence.
| Column Configuration | Ion Selectivity | Regeneration Cost | Operational Life |
|---|---|---|---|
| Single Bed Cation | Medium | Low | 5-7 Years |
| Mixed Bed Column | Very High | High | 4-6 Years |
| Chelating Column | Extreme | Medium | 6-8 Years |
| Weak Base Column | High | Very Low | 7-10 Years |
| Adsorption Column | Medium | Low | 5-8 Years |
| Specialized Hybrid | High | Medium | 8-12 Years |
The ideal flow rate depends on the resin type and the target ion. Generally, it is measured in Bed Volumes (BV) per hour. For most industrial ion exchange resin column applications, a flow rate of 8-40 BV/h is common. If the flow is too fast, you risk early breakthrough; if too slow, the production capacity drops. Proper calculation based on the resin's kinetics is essential for optimization.
Regeneration is triggered when the column reaches "breakthrough," meaning the target ions start appearing in the effluent. This is typically monitored using inline conductivity meters or specific ion probes. For a professional ion exchange resin column setup, we recommend setting a trigger point at 90% of the theoretical capacity to ensure no contaminated water escapes the system.
While the vessel is a container, the material compatibility is key. For example, using strong acids for regeneration in a low-grade steel column will cause rapid corrosion. Furthermore, the physical density of the resin must match the column's underdrain design to prevent the resin from migrating or clogging the laterals. Always match your resin choice with the vessel's material and internal support structure.
Channeling occurs when the liquid finds a path of least resistance through the resin bed, bypassing large sections of the media. This is usually caused by poor resin loading (voids), damaged internals, or air pockets trapped during the start-up phase. To prevent this in an ion exchange resin column, ensure a slow, controlled fill and perform regular backwashing to re-stratify the bed.
A lead-lag configuration (two columns in series) is vastly superior for critical applications. The first column (lead) does the heavy lifting, and the second (lag) acts as a safety polisher. When the lead column breaks through, the lag column continues to protect the system, allowing you to regenerate the lead column without stopping production. This maximizes resin utilization and guarantees consistent output quality.
Depending on the water quality and regeneration practices, resins typically last between 3 to 10 years. Organic fouling and oxidative degradation (especially from chlorine) are the primary causes of capacity loss. Implementing a pre-filtration stage or using carbon filters before the ion exchange resin column can significantly extend the resin's operational life.
The ion exchange resin column is far more than a simple piece of industrial equipment; it is a precision tool that enables the highest standards of purity and resource recovery across the globe. By integrating the correct resin chemistry—whether it be Strong Acid Cation, Chelating, or Mixed Bed—with a robust vessel design and automated control systems, industries can achieve unprecedented efficiency, reduce chemical waste, and ensure regulatory compliance in an increasingly strict environmental landscape.
Looking ahead, the convergence of smart sensing and advanced polymer science will continue to push the boundaries of what these columns can achieve. For companies seeking to optimize their water treatment or mineral recovery processes, the focus should shift toward holistic system design rather than just resin replacement. We encourage you to evaluate your current configurations to identify bottlenecks in flow or regeneration. Visit our website for expert guidance and high-performance resin solutions: www.lijiresins.com

