







Ion exchange resins used in water treatment are more than just a scientific process – they're the bedrock of safe, accessible water for billions. From ensuring the water flowing from your tap is potable to powering crucial industrial processes, these resins are silently working to solve some of the world’s most pressing challenges. Understanding their function and potential is increasingly vital, especially as global water scarcity becomes a more prominent concern. It’s not just about chemistry; it's about public health, economic stability, and environmental sustainability.
The global demand for clean water is exploding, driven by population growth, industrial expansion, and increasing awareness of water quality issues. The UN estimates that billions lack access to safely managed drinking water services, and the World Bank highlights the significant economic impact of water pollution. This creates a massive, and growing, need for effective water treatment solutions, where ion exchange resins used in water treatment are proving to be indispensable. It’s a field that requires constant innovation and a deep understanding of both the science and the practical realities of deployment.
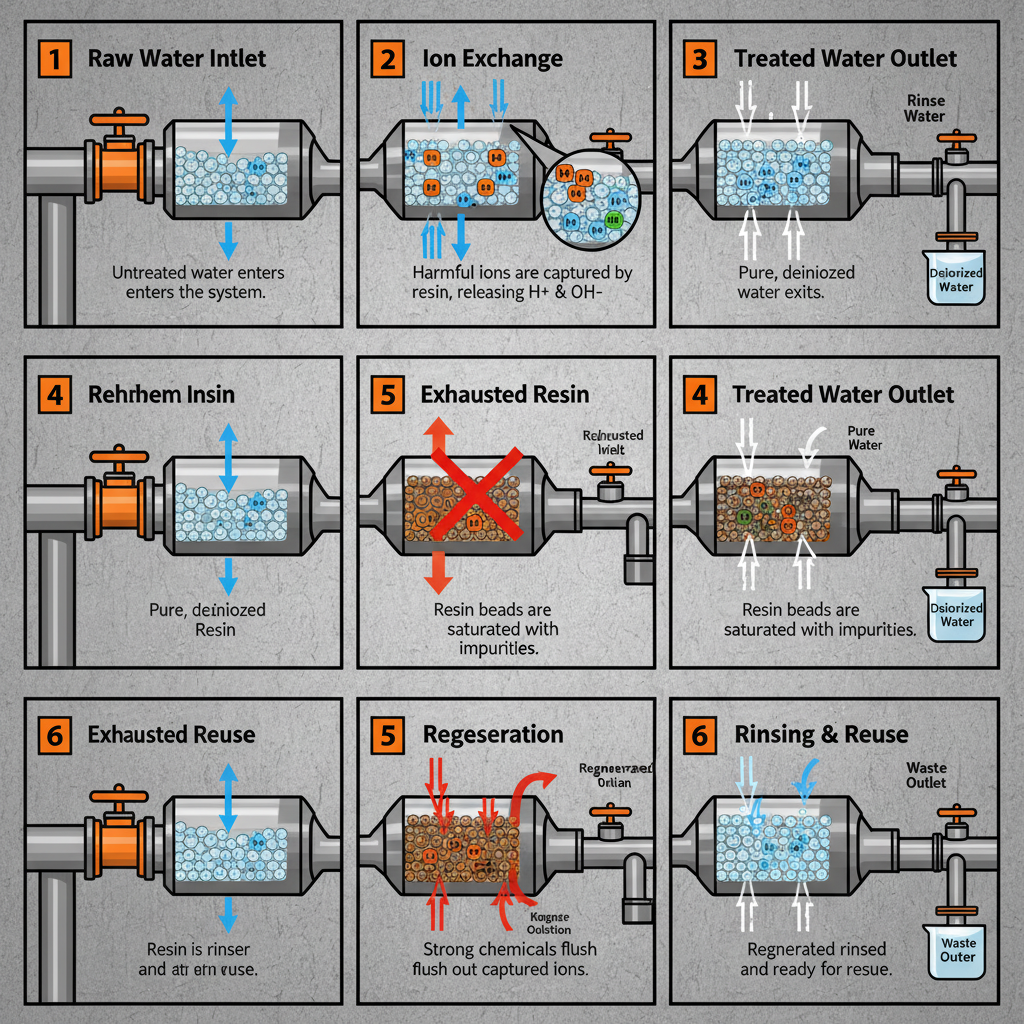
Simply put, ion exchange resins used in water treatment are specialized materials that swap unwanted ions in water with less harmful ones. Think of it like a molecular-level filter. They're typically small plastic beads with charged functional groups that attract and bind to specific ions, effectively removing contaminants like calcium, magnesium (hardness), nitrates, and even heavy metals. This isn’t some theoretical concept either, I’ve seen these resins in action on everything from municipal water plants to remote mining operations – they’re workhorses.

Look, you can get lost in the science of polymers and cross-linking, but at the end of the day, these resins are about solving real-world problems. I've been on sites where the water is so hard it clogs everything, and ion exchange is the only practical solution. It's not a glamorous fix, but it's reliable and crucial. We're talking about preventing scale buildup in boilers, softening water for laundry, and, most importantly, removing harmful contaminants from drinking water.
From a practical standpoint, the longevity of the resin is critical. You don’t want to be constantly replacing it. Factors like temperature, pH, and the concentration of contaminants all play a role. I’ve seen installations where the resin lasted for years with proper maintenance, and others where it needed replacement much sooner due to harsh conditions. That’s where understanding the application and selecting the right resin type really comes into play.
Ion exchange isn't a single process, it's a family of technologies. There are different resin types – strong acid cation, weak acid cation, strong base anion, weak base anion – each designed to target specific ions. Choosing the right one depends on the water chemistry and the contaminants you're trying to remove. It’s like picking the right tool for the job; you wouldn't use a hammer to tighten a screw.
The process itself involves passing water through a bed of these resin beads. The unwanted ions are attracted to the resin and displace the less harmful ions already bound to it. Once the resin is saturated, it needs to be regenerated – typically with a brine solution (saltwater) – to restore its capacity. That regeneration process is key, and where careful chemical management comes into play.
It's a vital component in modern water treatment, directly linked to the well-being of communities and industries. From a simple water softener in a home to complex industrial effluent treatment, ion exchange provides a reliable and cost-effective way to ensure water quality.
Durability is paramount. These resins take a beating, exposed to varying temperatures, pressures, and chemical compositions. You need resins that can withstand those conditions without degrading or losing their capacity. I've seen cheap resins break down quickly, leading to frequent replacements and increased costs.
Scalability matters. A solution that works for a small town might not be suitable for a large city or an industrial facility. You need resins that can be easily scaled up to meet varying demands without compromising performance. The ability to configure resin beds for different flow rates and contaminant levels is crucial.
Cost efficiency is always a factor. While initial cost is important, you need to consider the total cost of ownership – including regeneration chemicals, maintenance, and replacement frequency. A slightly more expensive resin that lasts longer and requires less maintenance can often be the more economical choice in the long run. And let's be real, budget constraints are always a factor.
The applications are incredibly diverse. You’ll find them in power plants removing impurities from boiler feedwater to prevent corrosion and scaling, in the food and beverage industry purifying water for production, and in pharmaceutical manufacturing ensuring water meets stringent purity standards. I’ve even seen them used in the nuclear industry for radioactive waste treatment.
In developing countries, ion exchange resins are critical for providing safe drinking water to communities lacking access to advanced treatment facilities. Small-scale, point-of-use systems utilizing these resins can dramatically improve public health. We've worked on projects in rural areas where a simple resin-based filter was the difference between sickness and health.
The semiconductor industry relies heavily on ultrapure water, and ion exchange is a key component in achieving that level of purity. Without it, you'd have defects in microchips, rendering them useless. It’s a high-stakes application where even the smallest contamination can have significant consequences.
Beyond just removing contaminants, ion exchange offers significant cost savings compared to some other treatment methods, especially when dealing with large volumes of water. Reducing scaling in industrial equipment alone can save companies a fortune in maintenance and downtime. It’s a proactive approach, preventing problems before they happen.
Sustainability is also a major benefit. By recovering valuable resources from wastewater – like metals – and reducing the need for harsh chemicals, ion exchange contributes to a more circular economy. It's not just about cleaning water; it's about responsible resource management.
We’re seeing a move towards more selective resins, designed to target specific contaminants with greater efficiency. Researchers are also exploring the use of bio-based resins – made from renewable resources – to further reduce the environmental impact. That's a big step forward.
Digitalization is playing an increasing role. Smart resin systems with real-time monitoring and automated regeneration are becoming more common, optimizing performance and reducing operational costs. The ability to remotely monitor and adjust treatment parameters is a game-changer, particularly for remote sites.
And let’s not forget the integration of ion exchange with other treatment technologies, like membrane filtration, to create hybrid systems that offer even greater performance and flexibility. It's about combining the best of different worlds.
Fouling – the buildup of organic matter and other debris on the resin surface – is a common challenge. It reduces resin capacity and requires more frequent cleaning. Pre-treatment of the water to remove these contaminants is crucial, but it adds to the overall cost. I’ve seen systems where inadequate pre-treatment led to complete resin failure.
The disposal of spent resin and regeneration byproducts is another concern. Finding environmentally sound ways to manage these materials is essential. Research into resin recycling and the development of more biodegradable resins are ongoing.
Maintaining consistent water quality is a constant battle. Fluctuations in contaminant levels can impact resin performance. Implementing robust monitoring systems and adjusting treatment parameters accordingly is vital. It’s a dynamic process that requires constant attention.
| Challenge | Impact | Potential Solution | Implementation Difficulty (1-5) |
|---|---|---|---|
| Resin Fouling | Reduced Capacity, Increased Cleaning | Enhanced Pre-Treatment Systems | 3 |
| Regeneration Byproduct Disposal | Environmental Concerns, Compliance Issues | Resin Recycling Programs | 4 |
| Water Quality Fluctuations | Inconsistent Treatment, Reduced Efficiency | Real-Time Monitoring & Control Systems | 2 |
| Resin Degradation | Reduced Lifespan, Frequent Replacement | Selection of Durable Resin Types | 2 |
| High Regeneration Chemical Costs | Increased Operational Expenses | Optimized Regeneration Cycles | 3 |
| Limited Selectivity for Complex Mixtures | Reduced Efficiency in Treating Multiple Contaminants | Hybrid Treatment Systems | 4 |
The lifespan varies significantly based on application, water quality, and resin type. Generally, high-quality resins can last 5-10 years with proper maintenance. Factors like extreme temperatures, high contaminant loads, and improper regeneration can shorten this lifespan. Regular monitoring of resin capacity is key to determining when replacement is necessary. Replacing worn-out resin ensures continued water quality and operational efficiency.
pH is a crucial factor. Cation resins generally work best in acidic conditions, while anion resins prefer alkaline environments. Extreme pH levels can damage the resin matrix, reducing its capacity and selectivity. Maintaining the optimal pH range for a specific resin type is essential for maximizing its effectiveness. Pre-treatment steps, like pH adjustment, are often necessary.
The most common regeneration chemical for cation resins is hydrochloric acid (HCl) or sulfuric acid (H2SO4), while sodium chloride (NaCl, or brine) is typically used for anion resins. The choice depends on the specific resin and the contaminants being removed. Proper handling and disposal of these chemicals are vital for safety and environmental compliance.
While ion exchange primarily targets dissolved ions, it doesn't effectively remove bacteria or viruses. It can reduce some organic matter that supports microbial growth, but disinfection methods (like UV sterilization or chlorination) are always necessary to ensure water is microbiologically safe. Ion exchange often forms part of a multi-barrier treatment system that includes disinfection.
The environmental impact primarily stems from the disposal of spent resin and the use of regeneration chemicals. Proper waste management practices and exploring biodegradable resin alternatives are crucial. Optimizing regeneration cycles to minimize chemical usage and investigating resin recycling options also reduce the environmental footprint.
Selecting the correct resin requires a detailed analysis of your water chemistry, the contaminants you need to remove, and the desired water quality. Consider factors like resin type (cation vs. anion, strong vs. weak), particle size, and capacity. Consulting with a water treatment specialist is highly recommended for optimal resin selection.
Ion exchange resins are a cornerstone of modern water treatment, offering a reliable, cost-effective, and versatile solution to a growing global challenge. From safeguarding public health to enabling critical industrial processes, their impact is profound and far-reaching. Understanding the nuances of these technologies – from resin selection to operational optimization – is vital for maximizing their benefits and ensuring sustainable water management.
Looking ahead, continued innovation in resin materials, coupled with the adoption of smart monitoring and control systems, will further enhance their performance and efficiency. Investing in research and development, promoting responsible waste management practices, and educating stakeholders on the importance of proper operation are crucial steps towards unlocking the full potential of ion exchange resins used in water treatment and securing a water-secure future for all.

