








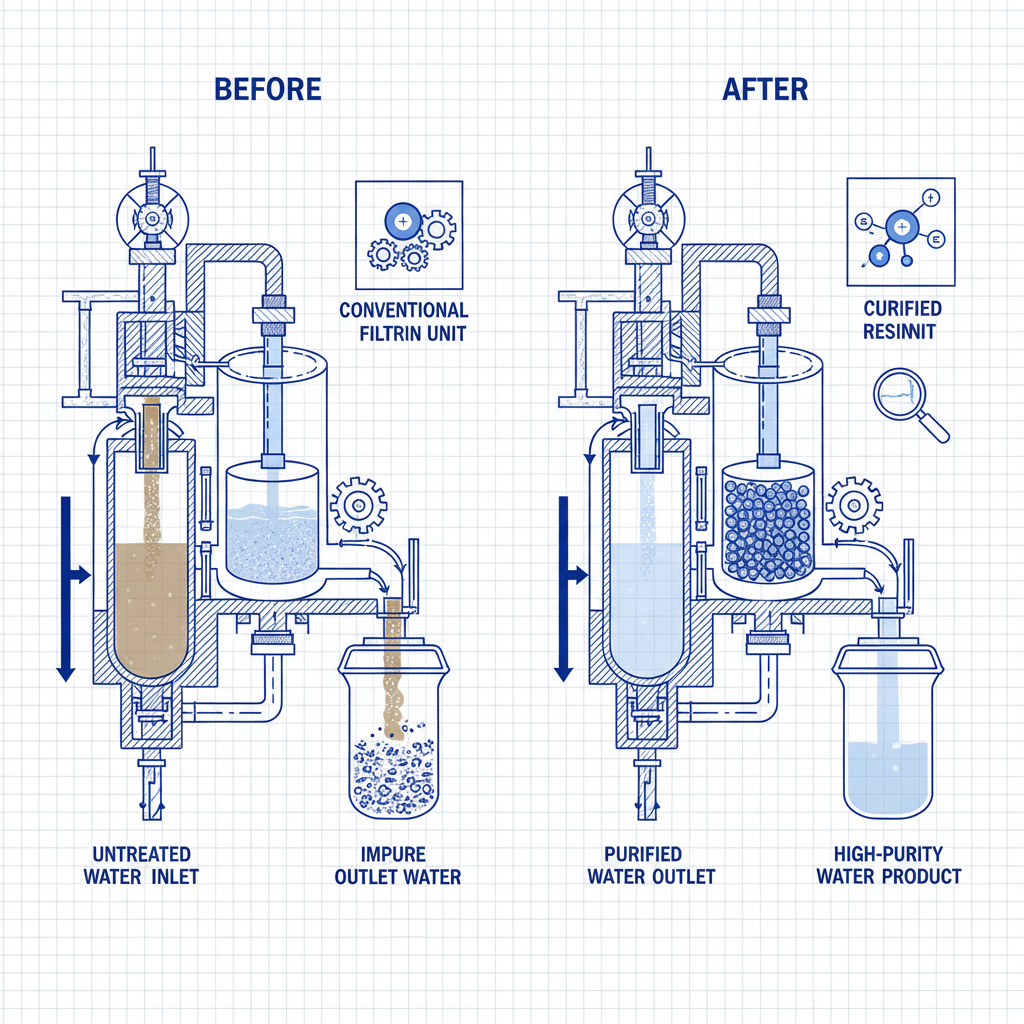
In the realm of water treatment and chemical processing, the role of cation exchange resin is absolutely fundamental. These specialized polymeric materials act as chemical "magnets," selectively removing positively charged ions from a solution and replacing them with others, typically hydrogen or sodium ions. Whether it is for softening hard water in industrial boilers or purifying pharmaceuticals, the efficiency of these resins determines the quality of the final output. Understanding how these resins function and how to choose the right type is essential for optimizing operational costs and ensuring environmental compliance. In this comprehensive guide, we will explore the science, types, and industrial impact of these powerful materials.

At its core, a cation exchange resin consists of a cross-linked polymer matrix—usually polystyrene—containing fixed negative functional groups. Because opposites attract, these negative sites hold onto positively charged ions (cations). When a liquid containing unwanted cations (such as Calcium Ca²⁺ or Magnesium Mg²⁺) passes through the resin bed, the resin swaps its preferred ion (like Na⁺) for the contaminant. This process continues until the resin sites are saturated, at which point the resin must be "regenerated" using a concentrated salt or acid solution to restore its capacity. This cyclical process makes ion exchange an incredibly sustainable and cost-effective method for large-scale water purification.
Pro Tip: The efficiency of ion exchange is heavily influenced by the cross-linking percentage of the resin; higher cross-linking typically increases physical stability but may slow down the exchange kinetics.
Not all resins are created equal. Depending on the chemical environment (specifically the pH level), engineers must choose between "Strong" and "Weak" variants. A strong cation exchange resin can remove ions across a wide pH range, making it the gold standard for complete demineralization. In contrast, weak resins are highly efficient but only operate within a narrow pH window, often used for removing alkalinity or specific heavy metals. Selecting the wrong type can lead to premature resin exhaustion or incomplete contaminant removal.
The versatility of cation exchange resin allows it to serve diverse sectors. In power plants, it prevents scale buildup in high-pressure boilers, which would otherwise lead to catastrophic equipment failure. In the food and beverage industry, it is used for sugar decolorization and water polishing. Furthermore, the mining industry utilizes these resins for the selective recovery of precious metals like gold and copper from leach solutions. By precisely tailoring the resin's pore size and functional group density, manufacturers can target specific ions while ignoring others, ensuring maximum purity.

To maximize the lifespan of your cation exchange resin, proper maintenance is non-negotiable. Fouling—the accumulation of organic matter or suspended solids on the resin beads—can block access to exchange sites, drastically reducing capacity. Implementing a robust pre-filtration system is the first line of defense. Additionally, monitoring the pressure drop across the resin bed can alert operators to bed compaction or clogging. Proper regeneration cycles, using the correct concentration of regenerants, ensure that the resin is fully "reset" without causing osmotic shock, which can crack the beads.
When procuring resin, technical data sheets provide the roadmap for system design. Key metrics include the Total Exchange Capacity (expressed in eq/L) and the Moisture Retention percentage. High-grade resins from Liji Resins are engineered for maximum durability and consistent bead size distribution to minimize pressure loss. Below is a typical specification table for a standard industrial-grade Strong Acid Cation resin:
Selecting the ideal cation exchange resin is a balance of chemistry, economics, and engineering. Whether your goal is simple water softening or complex industrial purification, the quality of the resin determines the stability of your process. By understanding the distinctions between strong and weak resins and maintaining a strict regeneration protocol, you can ensure long-term efficiency and high-purity results. For those seeking professional-grade solutions, partnering with a trusted supplier like Liji Resins ensures you receive products that meet global standards for capacity and durability.
The regeneration frequency depends on the "ionic load" of the influent water and the volume of the resin bed. For example, water with high calcium and magnesium levels will exhaust the resin faster. Most industrial systems use sensors to monitor the leakage of ions; once a specific threshold (breakthrough point) is reached, the system automatically triggers a regeneration cycle. Proper calculation of the exchange capacity ensures that you regenerate only when necessary, saving water and chemical costs.
While common salt (NaCl) is used, it is critical to use high-purity, pelletized salt. Impure salts can introduce insoluble minerals or organics that coat the cation exchange resin beads, leading to "fouling." This reduces the resin's ability to exchange ions and can cause pressure buildup in the vessel. For industrial applications, solar salt or refined vacuum salt is recommended to maintain the chemical integrity of the polymer matrix.
Typically, high-quality industrial resins last between 3 to 8 years. However, this lifespan is heavily influenced by the quality of the feed water and the precision of the regeneration process. Factors that shorten lifespan include exposure to strong oxidants (like chlorine), which break down the polymer cross-links, and organic fouling. Regular resin analysis and backwashing can help extend the life of the resin, ensuring you get the maximum return on your investment.
When resin beads fracture—often due to osmotic shock or mechanical stress—they create "fines." These tiny fragments fill the gaps between whole beads, increasing the pressure drop across the bed and potentially causing "channeling," where water bypasses large portions of the resin. This leads to premature ion leakage. Regular backwashing is essential to flush out these fines and maintain a uniform bed structure for optimal performance.